|
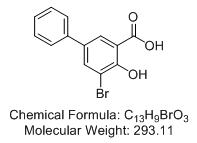
3-Bromo-5-phenylsalicylic acid (NSC109116)
• metabolism of progesterone in AKR1C1-overexpressed cells was potently inhibited by 3-bromo-5-phenylsalicylic acid, which was effective from 10 nM with an IC50 value equal to 460 nM
OTAVAchemicals Catalogue Number: 7070707133
CAS Registry Number: 4906-68-7
Purity: 95%+ (HPLC)
Ref.: El-Kabbani et al. Structure-Guided Design, Synthesis, and Evaluation of Salicylic Acid-Based Inhibitors Targeting a Selectivity Pocket in the Active Site of Human 20α-Hydroxysteroid Dehydrogenase (AKR1C1). Journal of Medicinal Chemistry (2009), 52, 3259-3264
Abstract: The first design, synthesis, and evaluation of human 20α-hydroxysteroid dehydrogenase (AKR1C1) inhibitors based on the recently published crystal structure of its ternary complex with inhibitor are reported. While the enzyme-inhibitor interactions observed in the crystal structure remain conserved with the newly designed inhibitors, the additional phenyl group of the most potent compound, 3-bromo-5-phenylsalicylic acid, targets a nonconserved hydrophobic pocket in the active site of AKR1C1 resulting in 21-fold improved potency (Ki = 4 nM) over the structurally similar 3α-hydroxysteroid dehydrogenase isoform (AKR1C2). The compound is hydrogen bonded to Tyr55, His117, and His222, and the phenyl ring forms additional van der Waals interactions with residues Leu308, Phe311, and the nonconserved Leu54 (Val in AKR1C2). Additionally, the metabolism of progesterone in AKR1C1-overexpressed cells was potently inhibited by 3-bromo-5-phenylsalicylic acid, which was effective from 10 nM with an IC50 value equal to 460 nM.
|
 HOME
HOME ABOUT
ABOUT
 SERVICES
SERVICES
 PRODUCTS
PRODUCTS
 Targeted Libraries
Targeted Libraries
 Biochemicals
Biochemicals
 RESEARCH
RESEARCH
 DOWNLOADS
DOWNLOADS ORDERING
ORDERING
 CONTACTS
CONTACTS

