|
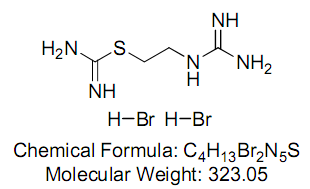
2-[(Aminoiminomethyl)amino]ethyl carbamimidothioic acid ester (S-(2-guanidylethyl)-isothiourea; VUF 8430 dihydrobromide)
VUF 8430 dihydrobromide is a potent histamine H4 receptor full agonist (pEC50 = 7.3), high affinity (pKi = 7.5)
OTAVAchemicals Catalogue Number: 7070707134
CAS Registry Number: 100130-32-3
CAS Registry Number: 98021-17-1 (free base)
Purity: 97%+
Ref. 1: Sterk et al. Studies on histaminergic compounds. IV. Non-isosterism between the imidazole, guanidino and isothiourea moieties at the H2-receptor. Archiv der Pharmazie (Weinheim, Germany) (1986), 319, 1057-1064
Abstract: S,S'-Alkylenediisothioureas were found to show only weak agonistic or antagonistic H2-activity, depending on the length of the alkylene chain. Introduction of a substituent in the amidino parts of both isothiourea groups resulted in a complete loss of the agonistic activity; only inactive compounds or weakly active H2-antagonists were obtained. Replacing one of the isothiourea groups of the alkylenediisothioureas with n = 2, 3 or 4 by a guanidino group has hardly any or no effect at all on the histamine H2-activity on the guinea-pig right atrium. However, extending the alkylene chain of these guanidylalkylisothioureas to n = 5 and 6 results in a strong increase in the H2-agonistic activity
Ref. 2: Lim et al. Pharmacological characterization of the new histamine H4 receptor agonist VUF
8430. British Journal of Pharmacology (2009), 157, 34-43
Background and purpose: We compare the pharmacological profiles of a new histamine H4 receptor agonist 2-(2-guanidinoethyl)isothiourea (VUF 8430) with that of a previously described H4 receptor agonist, 4-methylhistamine.
Experimental approach: Radioligand binding and functional assays were performed using histamine H4 receptors expressed in mammalian cell lines. Compounds were also evaluated ex vivo in monocyte-derived dendritic cells endogenously expressing H4 receptors and in vivo in anaesthetized rats for gastric acid secretion activity.
Key results: Both VUF 8430 and 4-methylhistamine were full agonists at human H4 receptors with lower affinity at rat and mouse H4 receptors. Both compounds induced chemotaxis of monocyte-derived dendritic cells. VUF 8430 also showed reasonable affinity and was a full agonist at the H3 receptor. Agmatine is a metabolite of arginine, structurally related to VUF 8430, and was a H4 receptor agonist with micromolar affinity. At histamine H3 receptors, agmatine was a full agonist, whereas 4-methylhistamine was an agonist only at high concentrations. Both VUF 8430 and agmatine were inactive at H1 and H2 receptors, whereas 4-methylhistamine is as active as histamine at H2 receptors. In vivo, VUF 8430 only caused a weak secretion of gastric acid mediated by H2 receptors, whereas 4-methylhistamine, dimaprit, histamine and amthamine, at equimolar doses, induced 2.5- to 6-fold higher output than VUF 8430.
Conclusions and implications: Our results suggest complementary use of 4-methylhistamine and VUF 8430 as H4 receptor agonists. Along with H4 receptor antagonists, both agonists can serve as useful pharmacological tools in studies of histamine H4 receptors.
Ref. 3: Lim et al. Discovery of S-(2-guanidylethyl)-isothiourea (VUF 8430) as a potent nonimidazole histamine H4 receptor agonist. Journal of Medicinal Chemistry (2006), 49, 6650-6651
Abstract: During an inhouse database screen, the authors identified S-(2-guanidylethyl)-isothiourea as a high affinity agonist for the histamine H4 receptor, with a 33-fold selectivity over the histamine H3 receptor and negligible affinity for the other histamine receptor subtypes. This nonimidazole ligand is introduced as a useful and complementary pharmacol. tool that enables further unraveling of the physiol. roles of the H4 receptor.
DOI:
10.1002/ardp.19863191202
10.1111/j.1476-5381.2009.00200.x
10.1021/jm060880d
Price info:
|
1 MG |
29 EUR |
|
5 MG |
49 EUR |
|
10 MG |
74 EUR |
|
300uL of 10mM solution |
39 EUR |
|
 HOME
HOME ABOUT
ABOUT
 SERVICES
SERVICES
 PRODUCTS
PRODUCTS
 Targeted Libraries
Targeted Libraries
 Biochemicals
Biochemicals
 RESEARCH
RESEARCH
 DOWNLOADS
DOWNLOADS ORDERING
ORDERING
 CONTACTS
CONTACTS

