|
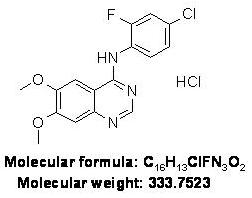
4-[(4'-Chloro-2'-fluoro)phenylamino]-6,7-dimethoxyquinazoline hydrochloride (ZM 306416)
OTAVAchemicals Catalogue Number: 7070707147
CAS Registry Number: 690206-97-4
Purity: 95%+ (HPLC)
Ref.: Hennequin et al Design and structure-activity relationship of a new class of potent VEGF receptor tyrosine kinase inhibitors. J.Med.Chem. 1999 42 5369.
Abstract: A series of substituted 4-anilinoquinazolines and related compounds were synthesized as potential inhibitors of vascular endothelial growth factor (VEGF) receptor (Flt and KDR) tyrosine kinase activity. Enzyme screening indicated that a narrow structure−activity relationship (SAR) existed for the bicyclic ring system, with quinazolines, quinolines, and cinnolines having activity and with quinazolines and quinolines generally being preferred. Substitution of the aniline was investigated and clearly indicated that small lipophilic substituents such as halogens or methyl were preferred at the C-4‘ position. Small substituents such as hydrogen and fluorine are preferred at the C-2‘ position. Introduction of a hydroxyl group at the meta position of the aniline produced the most potent inhibitors of Flt and KDR tyrosine kinases activity with IC50 values in the nanomolar range (e.g. 10, 12, 13, 16, and 18). Investigation of the quinazoline C-6 and C-7 positions indicates that a large range of substituents are tolerated at C-7, whereas variation at the C-6 is more restricted. At C-7, neutral, basic, and heteroaromatic side chains led to very potent compounds, as illustrated by the methoxyethoxy derivative 13 (IC50< 2 nM). Our inhibitors proved to be very selective inhibitors of Flt and KDR tyrosine kinase activity when compared to that associated with the FGF receptor (50- to 3800-fold). Observed enzyme profiles translated well with respect to potency and selectivity for inhibition of growth factor stimulated proliferation of human umbilical vein endothelial cells (HUVECs). Oral administration of selected compounds to mice produced total plasma levels 6 h after dosing of between 3 and 49 μM. In vivo efficacy was demonstrated in a rat uterine oedema assay where significant activity was achieved at 60 mg/kg with the meta hydroxy anilinoquinazoline 10. Inhibition of growth of human tumors in athymic mice has also been demonstrated: compound 34 inhibited the growth of established Calu-6 lung carcinoma xenograft by 75% (P < 0.001, one tailed t-test) following daily oral administration of 100 mg/kg for 21 days.
|
 HOME
HOME ABOUT
ABOUT
 SERVICES
SERVICES
 PRODUCTS
PRODUCTS
 Targeted Libraries
Targeted Libraries
 Biochemicals
Biochemicals
 RESEARCH
RESEARCH
 DOWNLOADS
DOWNLOADS ORDERING
ORDERING
 CONTACTS
CONTACTS

