|
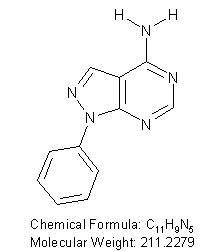
4-Amino-1-phenylpyrazolo[3,4-D]pyrimidine
Potent inhibitor of EGF-R PTK with IC50 = 2.7 μM.
OTAVAchemicals Catalogue Number: 0116690044
CAS Registry Number: 5334-30-5
Purity: 98%
Ref.: Traxler et al. Use of a Pharmacophore Model for the Design of EGF-R Tyrosine Kinase Inhibitors: 4-(Phenylamino)pyrazolo[3,4-d]pyrimidines J. Med. Chem. (1997), 40, 3601-3616.
Abstract: In the course of the random screening of a pool of CIBA chemicals, the two pyrazolopyrimidines 1 and 2 have been identified as fairly potent inhibitors of the EGF-R tyrosine kinase. Using a pharmacophore model for ATP-competitive inhibitors interacting with the active site of the EGF-R protein tyrosine kinase (PTK), the class of the pyrazolo[3,4-d]pyrimidines was then optimized in an interactive process leading to a series of 4-(phenylamino)-1H-pyrazolo[3,4-d]-pyrimidines as highly potent inhibitors of the EGF-R tyrosine kinase. The most potent compounds 13, 14, 15, 17, 19, 22, 26, 28, and 30 of this series inhibited the EGF-R PTK with IC50 values below 10 nM. High selectivity toward a panel of nonreceptor tyrosine kinases (c-Src, v-Abl and serine/threonine kinases (PKC R, CDK1) was observed. In cells, EGF-stimulated cellular tyrosine phosphorylation was inhibited by compounds 13, 15, 19, 22, and 23 at IC50values below 50 nM, whereas PDGF-induced tyrosine phosphorylation was not affected by concentrations up to 10 μM, thus indicating high selectivity for the inhibition of the ligand-activated EGF-R signal transduction pathway. Compounds 15 and 19 inhibited proliferation of the EGF-dependent MK cell line with IC50 values below 0.5 μM. In addition, two compounds, 9 and 11, showing satisfactory oral bioavailability in mice after oral administration, exhibited good in vivo efficacy at doses of 12.5 and 50 mg/kg in a nude mouse tumor model using xenografts of the EGF-R overexpressing A431 cell line. From SAR studies, a binding mode for 4-(phenylamino)-1H-pyrazolo[3,4-d]pyrimidines, especially for compound 15, at the ATP-binding site of the EGF-R tyrosine kinase is proposed. 4-(Phenylamino)-1H-pyrazolo[3,4-d]pyrimidines represent a new class of highly potent tyrosine kinase inhibitors which preferentially inhibit the EGF-mediated signal transduction pathway and have the potential for further evaluation as anticancer agents.
|
 HOME
HOME ABOUT
ABOUT
 SERVICES
SERVICES
 PRODUCTS
PRODUCTS
 Targeted Libraries
Targeted Libraries
 Biochemicals
Biochemicals
 RESEARCH
RESEARCH
 DOWNLOADS
DOWNLOADS ORDERING
ORDERING
 CONTACTS
CONTACTS

