|
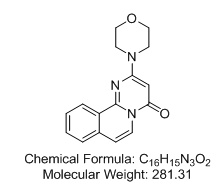
2-(4-Morpholinyl)-4H-Pyrimido[2,1-a]isoquinolin-4-one
Reversible and selective inhibitor of DNA-dependent protein kinase (DNA-PK) and mammalian target of rapamycin (mTOR) (IC50 values are 0.28 and 5.3 ìM respectively). Displays little affinity for other
commonly studied kinases including PI 3-K, ATM and ATR (IC50 values are all > 100 ìM). Induces apoptosis in vitro
OTAVAchemicals Catalogue Number: 7070707024
CAS Registry Number: 168425-64-7
Purity: 97%+ (HPLC)
Ref. 1: Ballou et al. Inhibition of Mammalian Target of Rapamycin Signaling by 2-(Morpholin-1-yl)pyrimido[2,1-a]isoquinolin-4-one. Journal of Biological Chemistry (2007), 282, 24463-24470
Abstract: Signalling through the mammalian target of rapamycin (mTOR) is hyperactivated in many human tumors, including hamartomas associated with tuberous sclerosis complex (TSC). Several small molecules such as LY294002 inhibit mTOR kinase activity, but they also inhibit phosphatidylinositol 3-kinase (PI3K) at similar concns. Compound 401 is a synthetic inhibitor of DNA-dependent protein kinase (DNA-PK) that also targets mTOR but not PI3K in vitro (Griffin, R. J., Fontana, G., Golding, B. T., Guiard, S., Hardcastle, I. R., Leahy, J. J., Martin, N., Richardson, C., Rigoreau, L., Stockley, M., and Smith, G. C. (2005) J. Med. Chem. 48, 569-585). Compound 401 was used to test the cellular effect of mTOR inhibition without the complicating side effects on PI3K. Treatment of cells with 401 blocked the phosphorylation of sites modified by mTOR-Raptor and mTOR-Rictor complexes (ribosomal protein S6 kinase 1 Thr389 and Akt Ser473, resp.). By contrast, there was no direct inhibition of Akt Thr308 phosphorylation, which is dependent on PI3K. Similar effects were also observed in cells that lack DNA-PK. The proliferation of TSC1-/- fibroblasts was inhibited in the presence of 401, but TSC1+/+ cells were resistant. In contrast to rapamycin, long-term treatment of TSC1-/- cells with 401 did not up-regulate phospho-Akt Ser473. Because increased Akt activity promotes survival, this may explain why the level of apoptosis was increased in the presence of 401 but not rapamycin. These results suggest that mTOR kinase inhibitors might be more effective than rapamycins in controlling the growth of TSC hamartomas and other tumors that depend on elevated mTOR activity.
Ref. 2: Griffin et al. Selective benzopyranone and pyrimido[2,1-a]isoquinolin-4-one inhibitors of DNA-dependent protein kinase: Synthesis, structure-activity studies, and radiosensitization of a human tumor cell line in vitro. Journal of Medicinal Chemistry (2005), 48, 569-585
Abstract: A diverse range of chromen-2-ones, chromen-4-ones, and pyrimidoisoquinolin-4-ones was synthesized and evaluated for inhibitory activity against the DNA repair enzyme DNA-dependent protein kinase (DNA-PK), with a view to elucidating structure-activity relationships for potency and kinase selectivity. DNA-PK inhibitory activity varied widely over the series of compounds evaluated (IC50 values ranged from 0.19 to >10 mM), with excellent activity being observed for the 7,8-benzochromen-4-one and pyrimido[2,1-a]isoquinolin-4-one templates. By contrast, inhibitors based on the benzochromen-2-one (coumarin) or 2-aryl-7,8-benzochromen-4-one (flavone) scaffolds were less potent. Crucially, these studies revealed a very constrained structure-activity relationship at the 2-position of the benzopyranone and pyrimido[2,1-a]isoquinolin-4-one pharmacophore, with only a 2-morpholino or 2-(2'-methylmorpholino) group being tolerated at this position. More detailed biological studies conducted with the most potent inhibitor NU7163 (IC50 = 0.19 mM) demonstrated ATP-competitive DNA-PK inhibition, with a Ki value of 24 nM, and NU7163 exhibited selectivity for DNA-PK compared with the related enzymes ATM, ATR, mTOR, and PI 3-K (p110alpha). NU7163 sensitized the HeLa human tumor cell line to the cytotoxic effects of ionizing radiation in vitro, a dose modification factor of 2.3 at 10% survival being observed with an inhibitor concentration of 5 mM. This study identified these structural classes as novel DNA-PK inhibitors and delineated initial structure-activity relationships against DNA-PK.
DOI:
10.1074/jbc.M704741200
10.1021/jm049526a
Price info:
|
1 MG |
59 EUR |
|
5 MG |
99 EUR |
|
10 MG |
129 EUR |
|
300uL of 10mM solution |
79 EUR |
|
 HOME
HOME ABOUT
ABOUT
 SERVICES
SERVICES
 PRODUCTS
PRODUCTS
 Targeted Libraries
Targeted Libraries
 Biochemicals
Biochemicals
 RESEARCH
RESEARCH
 DOWNLOADS
DOWNLOADS ORDERING
ORDERING
 CONTACTS
CONTACTS

